What are CYP450 and UGT enzymes?
Cytochrome P450 (CYP450) and UDP-glucuronosyltransferase (UGT) enzymes are major pathways in drug biotransformation and are involved in the metabolism of many psychotropic medications.1 Variations in these genes can lead to alterations in drug levels and overall drug exposure.
What are the functions of CYP450 enzymes?
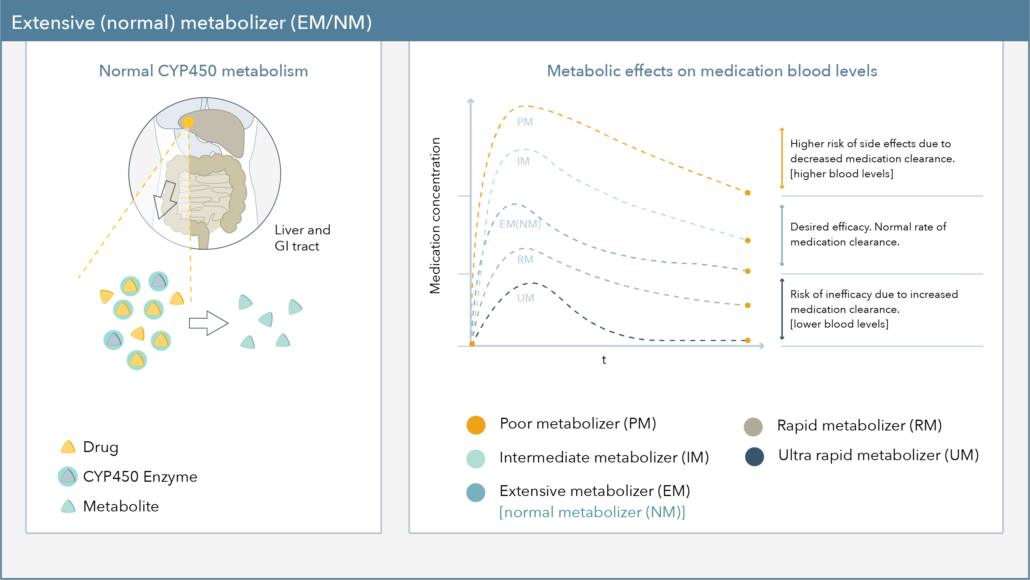
CYP450s are a superfamily of enzymes that play an important role in Phase I drug metabolism. These enzymes are present in several organs, including the liver, small intestine, kidneys, and lungs.2
CYP450s have been characterized as the main enzyme system for drug metabolism; in fact, 70-80% of medications are metabolized by CYP450s. Medications may be metabolized primarily by one enzyme or by the combined effect of multiple enzymes.3
The Genomind pharmacogenetic (PGx) report outlines genetic variants in six key CYP450 genes: 2D6, 2C9, 2C19, 2B6, 3A4/3A5, and 1A2.
Figure 1 demonstrates the effect genetic variations can have on CYP450 metabolism. Patients are typically reported as either extensive/normal (EM/NM), intermediate (IM), poor (PM), rapid (RM), or ultra-rapid metabolizers (UM).4 RMs and UMs, or fast metabolizers, are at risk for hyper-metabolism of medications, reduced blood levels, and may require a higher dose of medication to achieve therapeutic efficacy. IMs and PMs are at risk for slower drug metabolism, increased blood levels, and may require a reduced dose of medication to decrease the risk for side effects.5
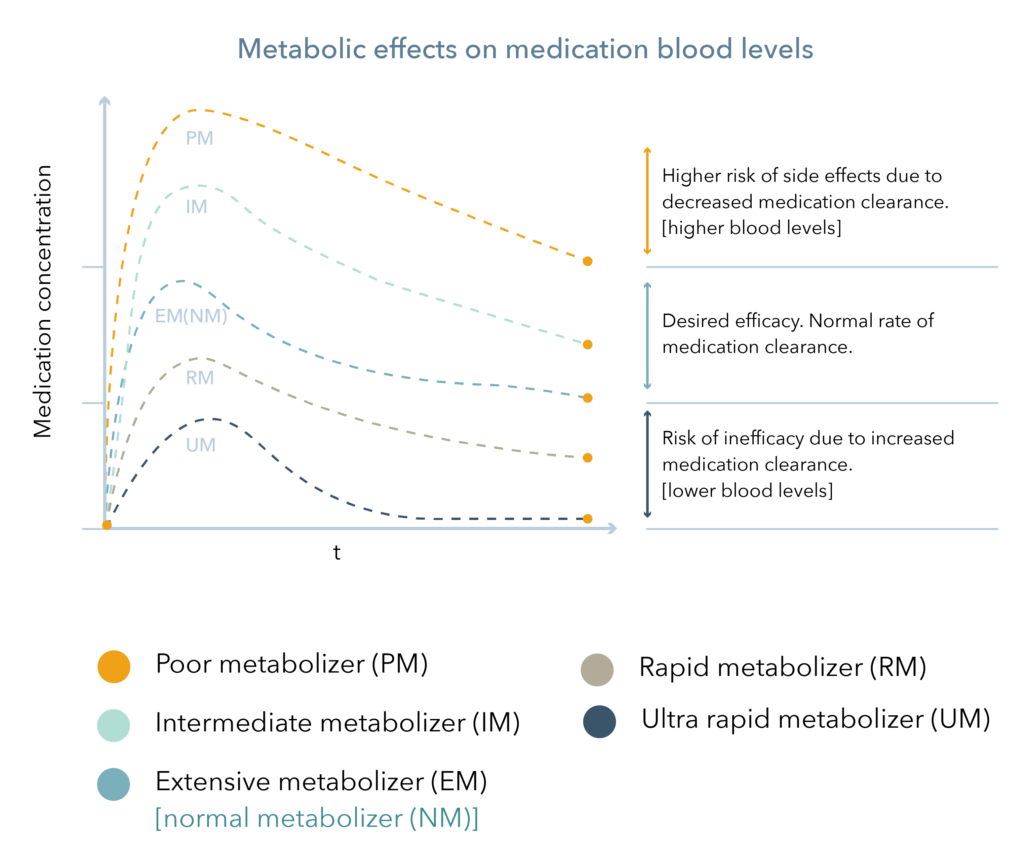
Figure 1. Metabolic Effects on Medication Blood Levels
Several psychotropic medications have specific guidance regarding the clinical effects of CYP450 variants. As an example, the FDA labeling for citalopram specifically cites “…in CYP2C19 poor metabolizers, citalopram steady state Cmax and AUC was increased by 68% and 107%, respectively. Citalopram [Celexa] 20 mg/day is the maximum recommended dose in CYP2C19 poor metabolizers due to the risk of QT prolongation…”6 Additional clinical guidance for CYP variants are provided by the Clinical Pharmacogenetics Implementation Consortium (CPIC) and The Dutch Pharmacogenetics Working Group (DPWG).
What are the functions of UGT enzymes?
UGT enzymes, on the other hand, are important components in Phase II metabolism of medications, which leads to the formation of more water-soluble and less toxic compounds that are more easily excreted.
UGT enzymes are found throughout several areas of the body, including the liver, skin, lungs, small intestine, and kidneys and are grouped into 2 subfamilies: UGT1 and UGT2.7
Genomind’s PGx test specifically reports on UGT1A4, which is involved in the biotransformation of lamotrigine, asenapine, and trifluoperazine, and UGT2B15, which metabolizes several benzodiazepine medications. Similar to the CYP450s, variants in the genes encoding for UGT can alter enzyme activity levels, which in turn can lead to variations in drug exposure. As an example, in a study of 24 healthy subjects, individuals with the decreased activity UGT2B15*2/*2 genotype were found to have a 0.58-fold lower clearance of intravenous lorazepam compared to those with the normal activity *1/*1 genotype.8
In Conclusion
Taken together, variants in CYP450 and UGT enzymes can provide clinicians with greater insight into a patient’s individual drug metabolism profile. This insight may allow for more patient-specific treatment decisions and medication choices.
Are You Ready to Upgrade Your Practice with Genomind?
Genomind’s pharmacogenetic testing is the most advanced and comprehensive mental health pharmacogenetic test available. Get access to 26 genes related to mental health, 130+ medications, 10+ conditions, state-of-the-art tools, and 360 degrees of support today!
References
- Lyubimov, A.V., Stingl, J., Oesterheld, J. and Turpeinen, M. (2012). Metabolism of Psychotropic Drugs. In Encyclopedia of Drug Metabolism and Interactions, A.V. Lyubimov (Ed.). Metabolism of Psychotropic Drugs – Stingl – Major Reference Works – Wiley Online Library
- Lynch T, Price A. The effect of cytochrome P450 metabolism on drug response, interactions, and adverse effects. Am Fam Physician 2007;76(3):391-396.
- Zanger UM, Schwab M. Cytochrome P450 enzymes in drug metabolism: regulation of gene expression, enzyme activities, and impact of genetic variation. Pharmacol Ther 2013;138(1):103-41.
- Gilani B, Cassagnol M. Biochemistry, Cytochrome P450. [Updated 2020 May 4]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2020 Jan. Available from: Biochemistry, Cytochrome P450 – StatPearls – NCBI Bookshelf (nih.gov)
- American Medical Association. Pharmacogenomics: Increasing the safety and effectiveness of drug therapy. Available at: https://crediblemeds.org/application/files/1216/1571/0481/pgx-brochure2011.pdf
- Celexa. Package insert. Forest Laborotories, Inc.; 2011.
- de Leon J. Gulcuronidation enzymes, genes and psychiatry. Int J Neuropsychopharmacol 2003;6(1):57-72.
- Chung J, Cho J, Yu K, et al. Effect of the UGT2B15 genotype on the pharmacokinetics, pharmacodynamics, and drug interactions of intravenous lorazepam in healthy volunteers. Clin Pharmacol Ther 2005;77(6):486-94.