What is HLA?
The human leukocyte antigen (HLA) genes are part of a larger cluster of genes known as the human major histocompatibility complex (MHC). The MHC is an essential part of the vertebrate immune system but is commonly referred to as HLA in humans because of how it was originally discovered. The MHC locus contains a variety of genes that are highly polymorphic.1
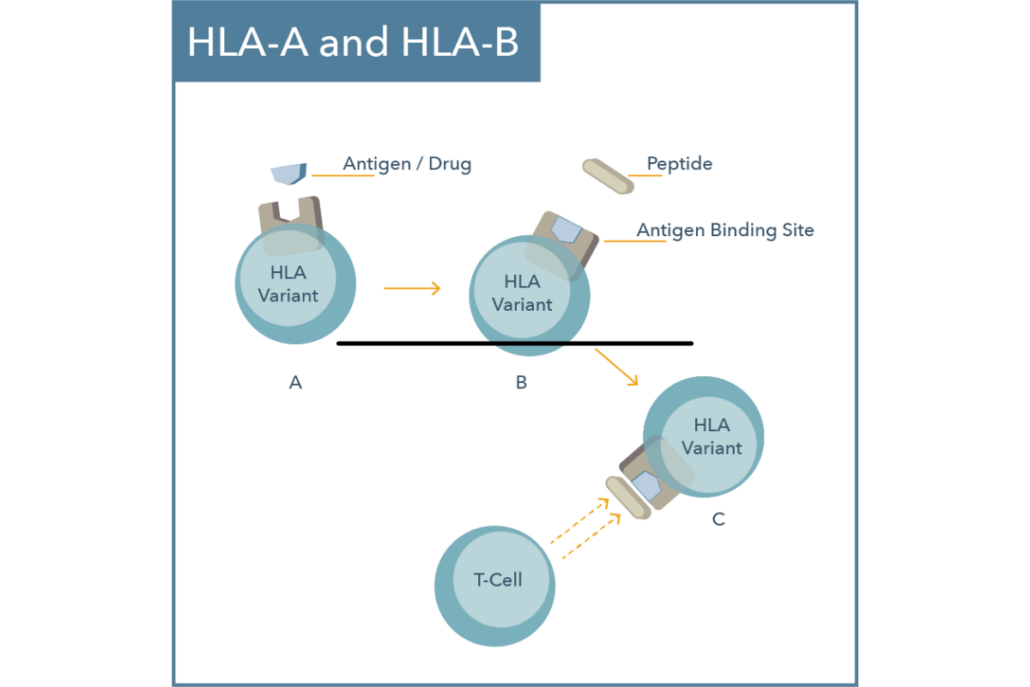
HLA itself is highly polygenic and is often divided into three categories: class I, II, and III. Each class has specific tissue locations and functions. The class I complex includes HLA-A, HLA-B, and HLA-C subtypes which encode cell surface proteins that present intracellular antigens to the immune system.1,2
These intracellular antigens are typically recognized as “self” as they are products of normal intracellular protein breakdown. However, if the presented antigen derives from a transplanted tissue or a pathogen, it may be recognized as “non-self” which could then trigger an immune response.2 Consequently, HLA gene variants have been associated with transplant rejection, autoimmune diseases, immune-mediated drug hypersensitivity reactions, and more.3
HLA Nomenclature
The World Health Organization (WHO) Nomenclature Committee for Factors of the HLA System is responsible for naming newly discovered HLA genes and allele sequences.4 According to the committee, there are more than 4,000 identified HLA-B alleles and more than 3,000 identified HLA-A alleles.2
Each HLA allele is assigned a unique name that contains at least a four digit identifier as shown in Figure 1. The first two digits provide information about the allele type while the second set of digits specify the protein subtype.2

Figure 1
As discussed in the article, Pharmacogenomic Terminology & Nomenclature, the Clinical Pharmacogenetics Implementation Consortium (CPIC) has taken on the responsibility of standardizing the field of pharmacogenetics, including terminology and also genotype to phenotype conversion tables. CPIC came to a consensus that HLA phenotypes would be organized by “high-risk genotype status” which is defined as follows:5
- Positive: detection of high-risk allele (homozygous or heterozygous for high-risk allele)
- Negative: high-risk allele not detected (no copies of high-risk allele)
Specific polymorphisms in the HLA gene have been associated with immune-mediated drug hypersensitivity reactions, including life-threatening cutaneous adverse reactions. As previously highlighted in our article Where Does Evidence for Pharmacogenetics Come From, CPIC curates and posts peer-reviewed, evidence-based, and detailed gene/drug clinical practice guidelines. To date, CPIC has published the following guidelines for various HLA gene-drug pairs:
- CYP2C9, HLA-B and Phenytoin
- HLA-A, HLA-B and Carbamazepine and Oxcarbazepine
- HLA-B and Abacavir
- HLA-B and Allopurinol
Cutaneous Adverse Drug Reactions
Several drugs are known to cause higher incidences of cutaneous adverse drug reactions (cADRs). These reactions can be life-threatening and are often unpredictable as they are independent of the prescribed dose.6 The clinical manifestations of cADRs include:2,7
- Maculopapular exanthema (MPE)
- Presence of rash (often small pink papules and maculae) without systemic features.
- Drug reaction with eosinophilia and systemic symptoms (DRESS)
- Generalized cutaneous eruptions with systemic manifestations that may be life-threatening.
- Stevens-Johnson syndrome (SJS)
- Severe blister-like rash involving the mucous membranes or organs and epidermal detachment < 10% of the body surface area. Mortality rates are typically below 5%.
- Toxic Epidermal Necrolysis (TEN)
- Severe blister-like rash involving the mucous membranes or organs and epidermal detachment > 30% of the body surface area. Mortality rates have been estimated to be as high as 30-50%.
Although life-threatening skin reactions are rare, it is important to address them due to their potential for severe consequences. HLA-B*15:02 and HLA-A*31:01 gene variants have been implicated in the development of life-threatening cADRs with several aromatic anticonvulsants that have indications for use in psychiatry.
HLA-B*15:02
HLA-B*15:02 is associated with an increased risk of skin reactions with carbamazepine, oxcarbazepine, lamotrigine, phenytoin, and fosphenytoin. Based on clinical data and/or similar structure; phenobarbital and eslicarbazepine may also be associated with increased risk of skin reactions in patients with this genotype. HLA-B*15:02 allele frequency varies by biogeographical group and is found most commonly in Asian countries.9
Carbamazepine and HLA-B*1502
Carbamazepine (Tegretol®, Equetro®), an antiepileptic drug, was prescribed nearly 3 million times in 2023.14 A systematic review published in 2012 by Yip and colleagues estimated that Asian patients carrying the HLA-B*1502 allele are approximately 113-times more likely to develop carbamazepine-induced SJS/TEN.15
Carbamazepine is one of the most common drugs associated with cADRs worldwide.6 SJS and TEN are estimated to occur in 1 to 6 per 10,000 new carbamazepine users in countries with predominantly Caucasian populations. However, the risk is estimated to be nearly 10 times higher in Asian countries where the high-risk allele (HLA-B*15:02) is inherited more frequently.8 (We expand on this idea further in our piece on ethnopsychopharmacology.)
Carbamazepine carries a boxed warning from the FDA which indicates that patients with ancestry in genetically at-risk populations should be screened for the presence of HLA-B*15:02 prior to initiating treatment with carbamazepine. “Patients testing positive for the allele should not be treated with carbamazepine unless the benefit clearly outweighs the risk.”8
As we mentioned earlier, CPIC has also published a guideline for carbamazepine use and HLA genotype. The guideline reviews both HLA-B*15:02 and HLA-A*31:01. If an individual is HLA-B*15:02 positive and is carbamazepine naïve, the guideline recommends not using carbamazepine. If the patient has previously used carbamazepine consistently for longer than three months without cutaneous adverse reactions, the guideline recommends that a clinician may cautiously consider using carbamazepine. The latency period for cADRs is variable, however, most reactions usually occur within three months of regular dosing.2
Oxcarbazepine and HLA-B*15:02
Oxcarbazepine (Trileptal®, Oxtellar®) is the keto-analog of carbamazepine and individuals carrying the HLA-B*15:02 allele may be at an increased risk for SJS/TEN with oxcarbazepine treatment.2 The FDA label states that testing should be considered prior to initiating treatment in patients with ancestry in genetically at-risk populations and that the use of oxcarbazepine should be avoided in patients who carry the HLA-B*15:02 allele unless the benefits clearly outweigh the risks.10 CPIC recommends that if a patient is oxcarbazepine-naïve and HLA-B*15:02 positive, do not initiate oxcarbazepine. If the patient has previously used oxcarbazepine consistently for longer than three months without cutaneous adverse reactions, the guideline recommends that a clinician may cautiously consider using oxcarbazepine.2
Phenytoin/Fosphenytoin and HLA-B*15:02
Phenytoin/fosphenytoin can lead to serious cADRs including SJS, TEN, and DRESS. According to the FDA Table of Pharmacogenomic Biomarkers in Drug Labeling, there is limited evidence suggesting that HLA-B*15:02 may be a risk factor for the development of SJS/TEN in patients taking phenytoin/fosphenytoin. According to the FDA, consideration may be given to avoiding phenytoin/fosphenytoin as an alternative to carbamazepine in patients who are positive for HLA-B*15:02.12 CPIC has also published a guideline for CYP2C9, HLA-B and Phenytoin which provides clinical recommendations for phenytoin/fosphenytoin based on CYP2C9 and HLA-B*15:02.11
Lamotrigine and HLA-B*15:02
According to the FDA label, the background rates of serious skin reactions are approximately 0.8% (8 per 1,000) in pediatrics and 0.3% (3 per 1,000) in adults receiving lamotrigine as adjunctive therapy for epilepsy.17 Lamotrigine may also be associated with increased risk of skin reactions in patients with HLA-B*15:02, however, the evidence is limited. A recent meta-analysis conducted by Deng and colleagues sought to investigate the association between HLA variants and lamotrigine-induced cADRs. The study found that HLA-B*15:02 carriers are approximately 2.4-times more likely to develop lamotrigine-induced SJS/TEN in Chinese populations.16
Other Anticonvulsants and HLA-B*15:02
Other aromatic anticonvulsants, including phenobarbital and eslicarbazepine, may also be associated with increased risk of skin reactions in patients with HLA-B*15:02. The evidence linking SJS/TEN with these agents is “very limited” according to CPIC. However, the CPIC recommendation is to use caution if initiating one of these alternative agents in an individual who is HLA-B*15:02 positive.2,11
HLA-A*31:01 and Carbamazepine
Compared to HLA-B*15:02, the HLA-A*31:01 allele is associated with a wider range of carbamazepine-induced cADRs (MPE, DRESS, SJS and TEN) in many different ethnicities.2 HLA-A*31:01 is associated with nearly a ten-fold increased risk of developing carbamazepine-induced hypersensitivity reactions whereas the HLA-B*15:02 is associated with a 113 fold increased risk.15 HLA-A*31:01 allele frequency varies by biogeographical group and is found most commonly in Caucasian individuals.9
The FDA Label for Carbamazepine states that the risks and benefits should be weighed before considering carbamazepine in patients known to be positive for HLA-A*31:01.8 As we mentioned earlier, the CPIC guideline for carbamazepine includes both HLA-B*1502 and HLA-A*31:01. If a patient is HLA-A*31:01 positive and carbamazepine-naïve, carbamazepine should be avoided if alternative agents are available. CPIC guidance notes that the other aromatic anticonvulsants have “very limited evidence, if any, linking SJS/TEN, DRESS, and/or MPE with the HLA-A*31:01 allele, and thus no recommendation can be made with respect to choosing another aromatic anticonvulsant as an alternative agent”. If no alternative agents are available, carbamazepine may be considered with increased clinical monitoring.2
Genomind’s Industry-Leading HLA-A Test Innovation13
A common industry approach for testing HLA-A*31:01 is using a surrogate marker known as a tag SNP. During a recent validation exercise, Genomind identified a 40% false positive rate with this method. Because of this, additional testing is recommended to confirm results of this method. However, Genomind scientists, led by the Chief Scientific Officer & Lab Director Dr. David Robbins, developed an innovative and proprietary real-time PCR (polymerase chain reaction) test that is highly specific for HLA-A*31:01. The increased specificity of this novel assay reduces the 40% false positive rate and renders confirmatory testing unnecessary. Genomind’s pharmacogenetic test is now the only mental health pharmacogenetics (PGx) test using this capability in the market.
Are You Ready to Upgrade Your Practice with Genomind?
Genomind’s pharmacogenetic testing is the most advanced and comprehensive mental health pharmacogenetic test available. Get access to 26 genes related to mental health, 130+ medications, 10+ conditions, state-of-the-art tools, and 360 degrees of support. Register today!
References
- Choo S. Y. (2007). The HLA system: genetics, immunology, clinical testing, and clinical implications. Yonsei medical journal, 48(1), 11–23. https://doi.org/10.3349/ymj.2007.48.1.11
- Phillips, E. J., Sukasem, C., Whirl-Carrillo, M., Müller, D. J., Dunnenberger, H. M., Chantratita, W., Goldspiel, B., Chen, Y. T., Carleton, B. C., George, A. L., Jr, Mushiroda, T., Klein, T., Gammal, R. S., & Pirmohamed, M. (2018). Clinical Pharmacogenetics Implementation Consortium Guideline for HLA Genotype and Use of Carbamazepine and Oxcarbazepine: 2017 Update. Clinical pharmacology and therapeutics, 103(4), 574–581. https://doi.org/10.1002/cpt.1004
- Martin MA, Hoffman JM, Freimuth RR, Klein TE, Dong BJ, Pirmohamed M, Hicks JK, Wilkinson MR, Haas DW, Kroetz DL; Clinical Pharmacogenetics Implementation Consortium. Clinical Pharmacogenetics Implementation Consortium Guidelines for HLA-B Genotype and Abacavir Dosing: 2014 update. Clin Pharmacol Ther. 2014 May;95(5):499-500. doi: 10.1038/clpt.2014.38. Epub 2014 Feb 21. PMID: 24561393; PMCID: PMC3994233.
- Robinson J, Halliwell JA, Hayhurst JH, Flicek P, Parham P, & Marsh SGE. The IPD and IMGT/HLA database: allele variant databases. Nucleic Acids Research (2015) 43:D423-431
- Caudle, K. E., Dunnenberger, H. M., Freimuth, R. R., Peterson, J. F., Burlison, J. D., Whirl-Carrillo, M., Scott, S. A., Rehm, H. L., Williams, M. S., Klein, T. E., Relling, M. V., & Hoffman, J. M. (2017). Standardizing terms for clinical pharmacogenetic test results: consensus terms from the Clinical Pharmacogenetics Implementation Consortium (CPIC). Genetics in medicine : official journal of the American College of Medical Genetics, 19(2), 215–223. https://doi.org/10.1038/gim.2016.87
- Mushiroda, T., Takahashi, Y., Onuma, T., Yamamoto, Y., Kamei, T., Hoshida, T., Takeuchi, K., Otsuka, K., Okazaki, M., Watanabe, M., Kanemoto, K., Oshima, T., Watanabe, A., Minami, S., Saito, K., Tanii, H., Shimo, Y., Hara, M., Saitoh, S., Kinoshita, T., … GENCAT Study Group (2018). Association of HLA-A*31:01 Screening With the Incidence of Carbamazepine-Induced Cutaneous Adverse Reactions in a Japanese Population. JAMA neurology, 75(7), 842–849. https://doi.org/10.1001/jamaneurol.2018.0278
- Zhang, J., Li, X., Su, Y., Sun, M., Wang, J., & Hao, Y. (2018). Association between HLA gene polymorphism and cutaneous adverse reactions caused by antiepileptic drugs. Experimental and therapeutic medicine, 15(4), 3399–3403. https://doi.org/10.3892/etm.2018.5801
- Tegretol. Package Insert. East Hanover, NJ: Novartis; 2009.
- Trileptal. Package Insert. East Hanover, NJ: Novartis; 2014.
- Karnes, J. H., Rettie, A. E., Somogyi, A. A., Huddart, R., Fohner, A. E., Formea, C. M., Ta Michael Lee, M., Llerena, A., Whirl-Carrillo, M., Klein, T. E., Phillips, E. J., Mintzer, S., Gaedigk, A., Caudle, K. E., & Callaghan, J. T. (2021). Clinical Pharmacogenetics Implementation Consortium (CPIC) Guideline for CYP2C9 and HLA-B Genotypes and Phenytoin Dosing: 2020 Update. Clinical pharmacology and therapeutics, 109(2), 302–309.
- U.S. Food and Drug Administration. Table of Pharmacogenomic Biomarkers in Drug Labeling. Available at: https://www.fda.gov/media/124784/download
- Genomind Announces Major Industry-Leading HLA-A Test Innovation on its Comprehensive Mental Health Pharmacogenetics (PGx) Test, Increasing Utility for Tens of Thousands of Clinicians and Patients. (2021). Retrieved 28 October 2021. Available at: https://genomind.com/press/genomind-announces-major-industry-leading-hla-a-test-innovation-on-its-comprehensive-mental-health-pharmacogenetics-pgx-test-increasing-utility-for-tens-of-thousands-of-clinicians-and-patients/
- Sean P. Kane, B. (2021). Carbamazepine – Drug Usage Statistics, ClinCalc DrugStats Database. Retrieved 3 October 2025. Available at: https://clincalc.com/DrugStats/
- Yip VL, Marson AG, Jorgensen AL, Pirmohamed M, Alfirevic A. HLA genotype and carbamazepine-induced cutaneous adverse drug reactions: a systematic review. Clin Pharmacol Ther. 2012;92(6):757-765.
- Deng, Y., Li, S., Zhang, L., Jin, H., & Zou, X. (2018). Association between HLA alleles and lamotrigine-induced cutaneous adverse drug reactions in Asian populations: A meta-analysis. Seizure, 60, 163–171.
- Lamictal. Package Insert. Research Triangle Park, NC: GlaxoSmithKline; 2021.