Many factors can influence the therapeutic efficacy of a drug, including pharmacokinetics, which refers to the passage of drugs into the body, through it, and out of the body.
Think of pharmacokinetics as a drug’s journey through the body, during which it passes through four different phases: absorption, distribution, metabolism, and excretion (ADME). The four steps are:
- Absorption: Describes how the drug moves from the site of administration to the site of action.
- Distribution: Describes the journey of the drug through the bloodstream to various tissues of the body.
- Metabolism: Describes the process that breaks down the drug.
- Excretion: Describes the removal of the drug from the body.
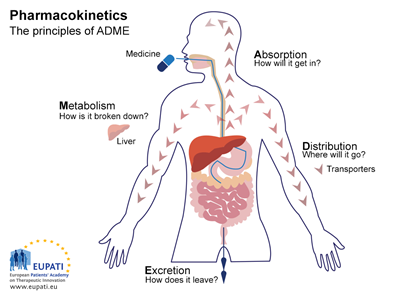
Let’s look at these phases in more detail:
Absorption
Absorption is the movement of a drug from its site of administration to the bloodstream. The rate and extent of drug absorption depend on multiple factors, such as:
- Route of administration
- The formulation and chemical properties of a drug
- Drug-food interactions
The administration (e.g., oral, intravenous, inhalation) of a drug influences bioavailability, the fraction of the active form of a drug that enters the bloodstream and successfully reaches its target site.
When a drug is given intravenously, absorption is not required, and bioavailability is 100% because the active form of the medicine is delivered immediately to the systemic circulation. However, orally administered medications have incomplete absorption and result in less drug delivery to the site of action. For example, many orally administered drugs are metabolized within the gut wall or the liver before reaching the systemic circulation. This is referred to as first-pass metabolism, which reduces drug absorption.
Distribution
The process of drug distribution is important because it can affect how much drug ends up in the active sites, and thus drug efficacy and toxicity. A drug will move from the absorption site to tissues around the body, such as brain tissue, fat, and muscle. Many factors could influence this, such as blood flow, lipophilicity, molecular size, and how the drug interacts with the components of blood, like plasma proteins.
For example, a drug like warfarin is highly protein-bound, which means only a small percentage of the drug is free in the bloodstream to exert its therapeutic effects. If a highly protein-bound drug is given in combination with warfarin, it could displace warfarin from the protein-binding site and increase the amount that enters the bloodstream.
Additionally, there are anatomical barriers found in certain organs like the blood-brain barrier, preventing certain drugs from going into brain tissue. Drugs with certain characteristics, like high lipophilicity, small size, and molecular weight will be better able to cross the blood brain barrier.
Metabolism
Cytochrome P450 (CYP450) enzymes are responsible for the biotransformation or metabolism of about 70-80% of all drugs in clinical use.
What are some factors that affect drug metabolism?
- Genetics can impact whether someone metabolizes drugs more quickly or slowly.
- Age can impact liver function; the elderly have reduced liver function and may metabolize drugs more slowly, increasing risk of intolerability, and newborns or infants have immature liver function and may require special dosing considerations.
- Drug interactions can lead to decreased drug metabolism by enzyme inhibition or increased drug metabolism by enzyme induction.
Generally, when a drug is metabolized through CYP450 enzymes, it results in inactive metabolites, which have none of the original drug’s pharmacologic activity. However, certain medications, like codeine, are inactive and become converted in the body into a pharmacologically active drug. These are commonly referred to as prodrugs.
As you can imagine, having genetic variations in CYP2D6, the metabolic pathway for codeine, can have significant clinical consequences. Usually, CYP2D6 poor metabolizers (PMs) have higher serum levels of active drugs. In codeine, PMs have higher serum levels of the inactive drug, which could result in inefficacy. Conversely, ultra-rapid metabolizers (UMs) will transform codeine to morphine extremely quickly, resulting in toxic morphine levels.
The FDA added a black box warning to the codeine drug label, stating that respiratory depression and death have occurred in children who received codeine following a tonsillectomy and/or adenoidectomy and who have evidence of being a CYP2D6 UM.

Excretion
Elimination involves both the metabolism and the excretion of the drug through the kidneys, and to a much smaller degree, into the bile.
Excretion into the urine through the kidneys is one of the most important mechanisms of drug removal.
Many factors affect excretion, such as:
- Direct renal dysfunction, which could prolong the half-life of certain drugs and necessitate dose adjustments.
- Age, which can contribute to differing rates of excretion and impact dosing of medications.
- Pathologies that impact renal blood flow, such as congestive heart failure and liver disease can make drug excretion less efficient
Whether it’s a patient who just had gastric bypass surgery, a CYP2D6 poor metabolizer, or a patient with renal dysfunction, an individual’s characteristics affect these four processes, which can ultimately influence medication selection.
In conclusion
The world of pharmacokinetics is vast, but understanding the basic mechanisms that govern the pharmacokinetics of a drug is vital to designing individualized treatment regimens for patients.
Pharmacogenetic testing with Genomind covers 11 pharmacokinetic genes that affect drug exposure and may inform drug dosage. Ask about pharmacokinetic genes during your next consultation!
Deliver targeted and personalized care with Genomind.
Register with Genomind to use our precision tools and services and help your patients get better. Get started today.
References
- Sakai JB. Pharmacokinetics: The Absorption, Distribution, and Excretion of Drugs. In: Practical Pharmacology for the Pharmacy Technician. 2009:27-40.
- Doogue MP. Polasek TM. The ABCD of clinical pharmacokinetics. Ther Adv Drug Saf. 2013;4(1):5-7.
- Fender AC and Dobrev D. Bound to bleed: how altered albumin binding may dictate warfarin treatment outcome. Int J Cardiol Heart Vasc. 2019;22:214-215.
- Banks WA. Characteristics of compounds that cross the blood-brain barrier. BMC Neurol. 2009;9(Suppl 1):S3.
- Zanger UM. Schwab M. Cytochrome P450 enzymes in drug metabolism: regulation of gene expression, enzyme activities, and impact of genetic variation. Pharmacol Ther. 2013;138(1):103-41.
- Ortiz deMontellano PR. Cytochrome P450-activated prodrugs. Future Med Chem. 2013;5(2):213-228.
