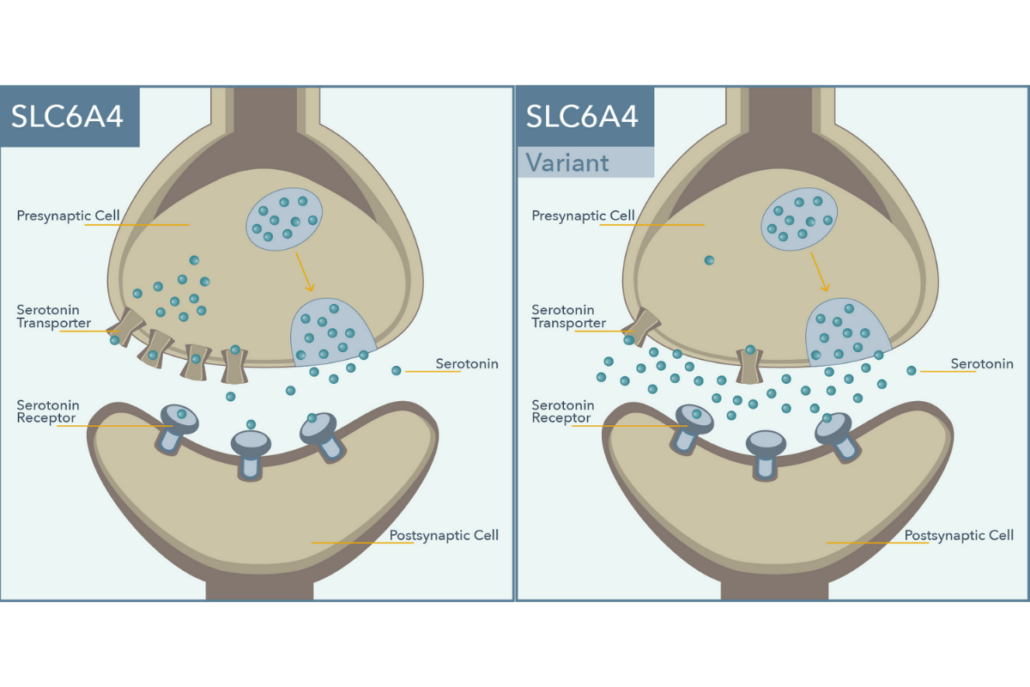
The SLC6A4 gene, or the solute carrier family 6, member 4 gene, encodes for a protein that transports serotonin from the synapse back into the neuron to halt signaling. The antidepressant activity of selective serotonin reuptake inhibitors (SSRIs) is achieved through inhibition of this transporter, allowing serotonin to remain in the synapse longer.1 A polymorphism within the promoter of this gene, i.e. 5-HTTLPR is a 44 base pair deletion that influences production of the serotonin transporter (SERT), with the long (L) allele associated with twice the basal expression as the short (S) allele. Another variant identified within the L allele (rs25531A/G) causes impaired expression similar to the S variant.2 Pharmacogenetic testing from Genomind analyzes these variations in SLC6A4 (See Figure 1), with a focus on their relationship with SSRI response.
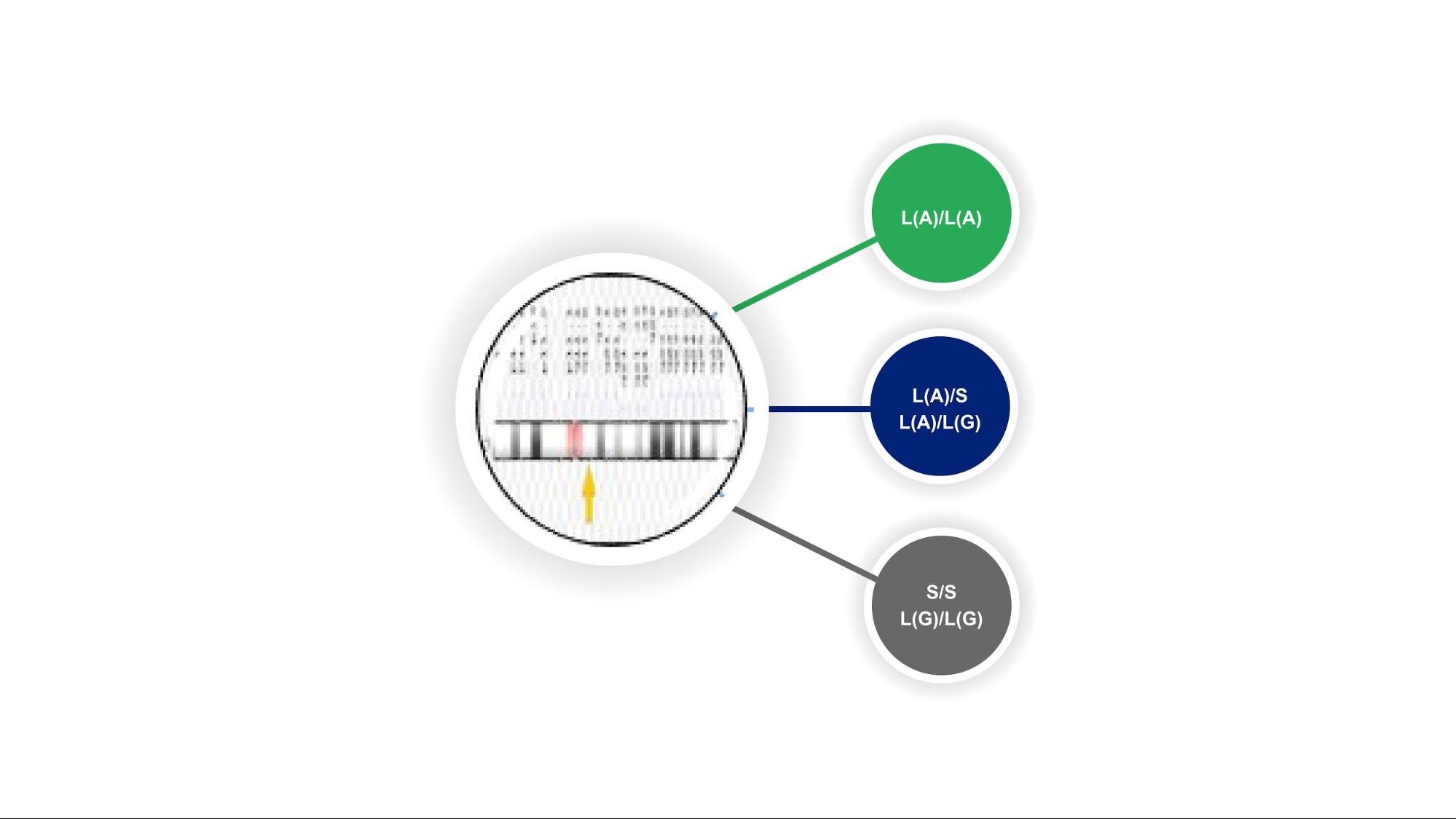
Figure 1. demonstrates the different genotypes that individuals may have based on variations in SLC6A4
A meta-analysis in Caucasian patients with depressive disorders showed that individuals with the S/S genotype were significantly less likely to respond to SSRIs than those with the L/L genotype (OR=1.71, p=0.003)3. This is consistent with results from a previous meta-analysis.4 Individuals who carry only a single risk variant (S or L(G) alone) appear to be at increased risk of adverse effects, particularly gastrointestinal side effects. While individuals with only a single risk allele have increased risk of adverse effects, those who carry two risk alleles [(S/S, S/L(G), and L(G)/L(G)] have greater risk of non-response and adverse effects.
We have collected a number of case studies that support the clinical utility of SLC6A4. In one case, a 30-year-old Caucasian female presented with intractable depression and severe anxiety along with a history of failures with a number of SSRIs. Genetic testing revealed her to have the short/short or S/S genotype, which is relevant to her history of SSRI failures. The antidepressant mirtazapine (15mg) was initiated at bedtime and then titrated up to 45mg. Mirtazapine was chosen since its mechanism is independent of serotonin transporter function. The patient reported improved mood and has been able to attend work consistently and exercise daily. Her affect and energy both improved. In addition, her Global Assessment of Functioning (GAF) increased from 20 to 60. This is one of many examples which demonstrate the utility of genetic information to guide treatment and improve outcomes in patients.
Are You Ready to Upgrade Your Practice with Genomind?
Genomind’s pharmacogenetic testing is the most advanced and comprehensive mental health pharmacogenetic test available. Get access to 26 genes related to mental health, 130+ medications, 10+ conditions, state-of-the-art tools, and 360 degrees of support today!
References
- Schloss P and Williams DC. The serotonin transporter: a primary target for antidepressant drugs. J Psychopharmacol. 1998;12(2):115-12.
- Ren F, Ma Y, Zhu X, Guo R, Wang J, He L. Pharmacogenetic association of bi- and triallelic polymorphisms of SLC6A4 with antidepressant response in major depressive disorder. J Affect Disord. 2020 Aug 1;273:254-264. doi: 10.1016/j.jad.2020.04.058. Epub 2020 May 11. PMID: 32421611. <https://pubmed.ncbi.nlm.nih.gov/32421611/>
- Porcelli S, Fabbri C, Serretti A. Meta-analysis of serotonin transporter gene promoter polymorphism (5-HTTLPR) association with antidepressant efficacy. Eur Neuropsychopharmacol. 2012;22(4):239-58.
- Serretti, A, Kato M, De Ronchi D, Kinoshita T. Meta-analysis of serotonin transporter gene promoter polymorphism (5-HTTLPR) association with selective serotonin reuptake inhibitor efficacy in depressed patients. Mol Psychiatry. 2007;12(3):247-57.