In recent years, the understanding of the role genetics plays in mental health has grown exponentially. With that understanding has come the ability for genetic testing to provide answers to questions that patients and their healthcare providers have about personalized treatment options.
Mental health is a function of genetics, environment, lifestyle and experiences – that’s the Mental Health 360 approach. Until now, we haven’t been able to fully access an important piece of this puzzle when it comes to mental health drug response for mental health conditions: your patient’s genetics.
The Mental Health 360
- Genetics – does not change
- Family history
- Genetic predispositions
- Brain chemical balance
- Metabolism
- Response to meds (PGx)
- Environment – variable
- Home and family life
- Socio-economic status
- Community
- Essential living conditions
- Education
- Lifestyle – variable
- Substance abuse
- Eating / sleeping behaviors
- Physical fitness
- Social life
- Employment
- Experience – variable
- Success and failure
- Abuse / trauma
- Childbirth
- Divorce
- Bullying
- Combat
Advancements in pharmacogenomics, optimized for mental health by Genomind over the last decade, have helped to unlock this information. Now healthcare providers can get a more complete understanding of their patient to identify a more personalized treatment plan.
What is pharmacogenomics?
Pharmacogenomics is a field of study combining the science of pharmacology and genomics; exploring how an individual’s genetics may influence their response to drugs. As we learned previously in our Introduction to Genetics:
“Different versions of the same gene referred to as different alleles or variants, and different combinations of alleles inherited from an individual’s parent’s account for most of the variation observable from person to person.”
This same concept is also applicable to our response to medications, and pharmacogenetic (PGx) testing may provide additional evidence and insight into medication response variability. PGx is one aspect of precision medicine – a model that emphasizes individualized care plans based on specific patient characteristics, such as one’s genotype.
PGx testing cannot determine “the perfect drug.” However, in conjunction with clinical judgment and reviewing your patient’s current symptoms, past treatment response, family history and treatment goals, clinicians can utilize PGx to make more robust and informed treatment decisions for their patients.
Genomind PGx test overview

Genomind’s PGx testing analyzes 26 selected genes that have been shown in numerous clinical studies to have implications for response and/or tolerability to treatments used for depression, anxiety, OCD, ADHD, bipolar disorder, and schizophrenia. These 26 genes are split into the two following categories.
Pharmacodynamics – what drugs do to our body
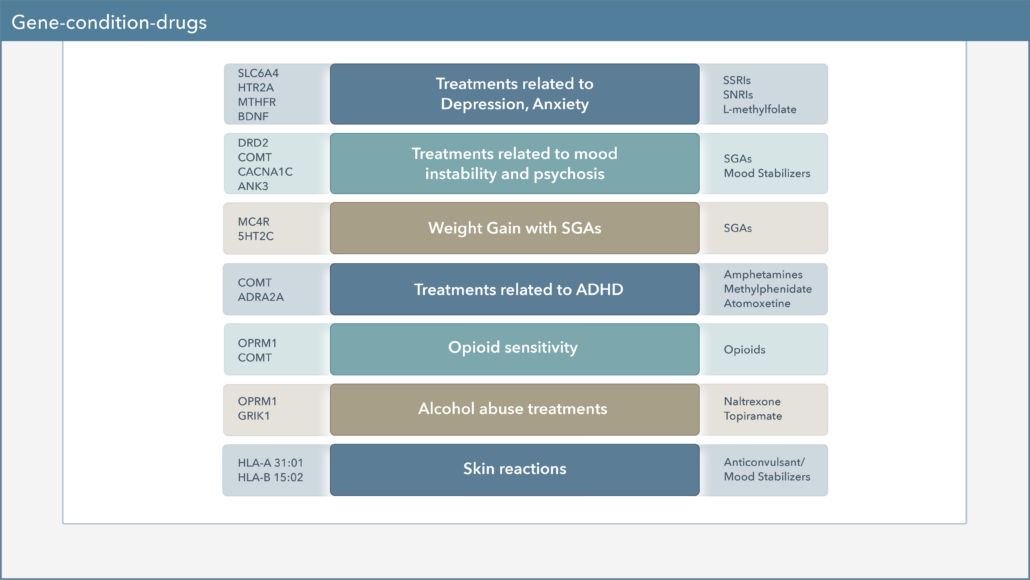
Our report’s pharmacodynamic section is a set of genes that can indicate the effect a drug has on the body and includes interactions with receptors, transporters, and neurotransmitters.
Pharmacokinetics – what our body does to drugs
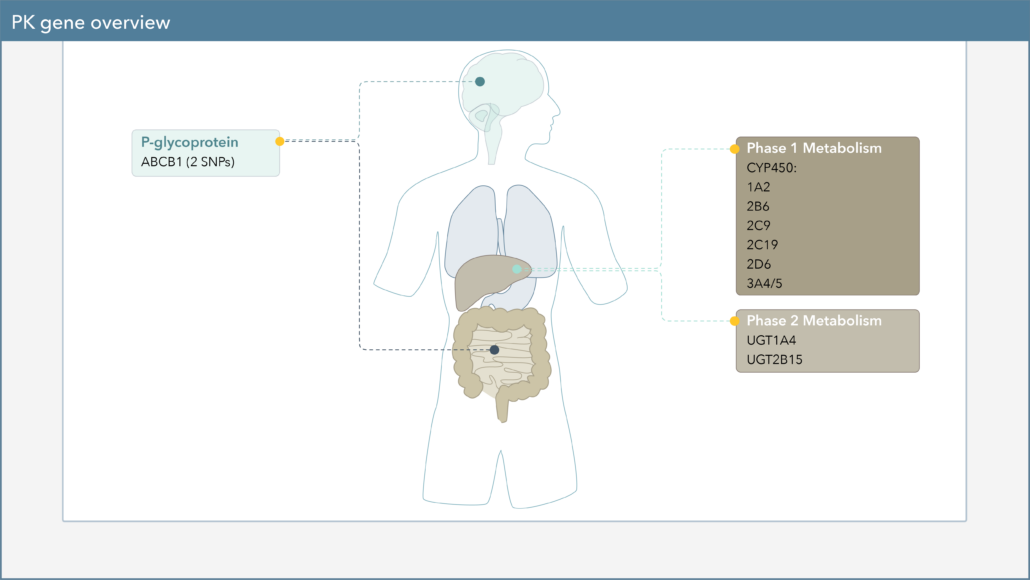
The pharmacokinetics section of the report includes genes related to the effect our body has on drugs, such as drug metabolism and absorption. The cytochrome P450 enzymes that we looked at in the section of Four Steps in a Drug’s Journey through the Body are in this part of the report.
Learn more about CYP genes and pharmacokinetics in our Pharmacokinetics (PK) and CYP450 Genes Introduction. Learn more about phenoconversion in our Introduction to Phenoconversion.
Evidence supporting pharmacogenetics
With pharmacogenetic testing being one of the newer tools clinicians have available at their disposal, a common question is what is the evidence behind all of this? Here is a snapshot of the evidence and organizations that support pharmacogenomics:
 The U.S. Food and Drug Administration (FDA) Table of Pharmacogenetic Associations and Table of Pharmacogenetic Biomarkers in Drug Labeling, includes 270+ drugs with pharmacogenetic precautions, warnings or dosing guidance on the label, including 36+ psychiatric drugs included on Genomind’s PGx report.
The U.S. Food and Drug Administration (FDA) Table of Pharmacogenetic Associations and Table of Pharmacogenetic Biomarkers in Drug Labeling, includes 270+ drugs with pharmacogenetic precautions, warnings or dosing guidance on the label, including 36+ psychiatric drugs included on Genomind’s PGx report.

Consortiums of researchers with the goal of developing PGx-based treatment guidelines, such as the Clinical Pharmacogenetics Implementation Consortium (CPIC) and the Dutch Pharmacogenetics Working Group (DPWG), are considered the leading authorities on standardizing pharmacogenetic practice. CPIC for example has produced peer reviewed pharmacogenetic guidelines on the SSRI’s, TCA’s, and atomoxetine.
 PharmGKB is a comprehensive pharmacogenomics database funded by the NIH, that curates knowledge about the impact of genetic variation on drug response and turns them into resources (drug label annotations, clinical guidelines, gene-drug associations and genotype-phenotype relationships) for clinicians and researchers.
PharmGKB is a comprehensive pharmacogenomics database funded by the NIH, that curates knowledge about the impact of genetic variation on drug response and turns them into resources (drug label annotations, clinical guidelines, gene-drug associations and genotype-phenotype relationships) for clinicians and researchers.
In addition to guidelines established by the FDA, CPIC, and DPWG the majority of pharmacodynamic and pharmacokinetic genes on the Genomind PGx test are supported by at least one of the following types of primary peer-reviewed publications: meta-analyses, systematic literature reviews, genome-wide association studies.
We also have our own set of published clinical evidence that supports the use of the Genomind PGx test. Brennan et al. (2015) found that 87% of patients utilizing Genomind PGx testing showed significant decreases in depression, anxiety, and medication side effects, and increased quality of life over 3 months. Another study conducted by Perlis et al. (2018) examining health care utilization and cost demonstrated that on average patients using Genomind PGx testing have a ~$2,000 reduction in health care costs in 6-months, 40% reduction in ER visits, and a 60% reduction in hospitalizations.
Most recently we have published our own randomized clinical trial conducted by Perlis et al. (2020). Although no significant differences were found within the primary outcome, exploratory analyses suggested significantly fewer individuals experienced worsening of depressive symptoms following Genomind PGx testing compare to treatment-as-usual, and that individuals receiving treatment that was concordant with PGx recommendations were twice (OR 2.23; 95% Cl 1.17-2.83) as likely to remit compared to patients who received non-concordant treatment.
For additional information on pharmacogenetic evidence, refer to our article on Where Does Evidence for Pharmacogenetics Come From.
In conclusion
Genomind’s platform of services is built around assisting clinicians with navigating treatment difficulties in mental health by providing a more complete picture of the patient.
In many instances, the practice of psychiatry can be challenging due to the common variability in treatment response. As a result, many clinicians and patients are often left with a trial-and-error approach, even when utilizing clinical guidelines. From a patient perspective, it can be frustrating to try treatment after treatment to only experience inefficacy or intolerable side effects.
Every clinician hopes to reduce the trial and error approach and to choose treatment plans that are better tailored to each individual patient. Evaluating genes in patients struggling with mental health conditions is a step towards achieving that goal.
Deliver targeted and personalized care with Genomind.
Register with Genomind to use our precision tools and services and help your patients get better. Get started today!
